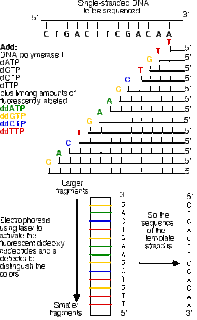
Most of us are familiar with the use of DNA profiling in criminal cases and with the establishment of the National DNA Database as an investigative tool on the back of this. DNA profiling can provide a high degree of confidence that a particular body fluid stain, hair or flake of dandruff, for instance, could have been deposited by one particular person. Indeed, a substantial part of Forensic Access’s work involves checking the accuracy and relevance of DNA test results and assessing the sustainability of conclusions drawn in respect of them as to likely culpability in specific case circumstances.
But the same sorts of techniques can have much wider applications, and these are discussed in this issue of Benchmark.
Establishing paternity
DNA analysis has been used in numerous paternity cases and is usually pretty straightforward. But one of the most challenging cases we have dealt with dates back to the 1950s, and has extended the frontiers of forensic DNA analysis.
The case concerned a man who, relatively late in life, discovered that the man that he had always thought was his father might not have been. Both his mother and his putative father were dead by the time his suspicions were aroused and he had no real clues to go on. One possibility for his paternity appeared to be a man living overseas with whom his mother had corresponded. Analysis of DNA in saliva on the backs of postage stamps on mail found amongst his mother’s possessions had the potential to hold the key – saliva that had been deposited more than 50 years ago!
Standard techniques produced weak, incomplete profiles of the DNA. It was not possible to exclude either of the men on the basis of these profiles, so we decided to attempt a new form of profiling that analyses DNA on the male-specific Y chromosome only. Y-chromosome profiles are passed on pretty much intact from father to son for generation after generation and so can provide a particularly powerful means of establishing paternity. In this case, the Y-chromosome profiling showed that the recent information was incorrect and indicated strongly that his father had been the man he always thought he was.
Identifying bodies or body parts
Over the years we have been increasingly successful in establishing the identity of human remains by applying our ever more sensitive DNA extraction and profiling techniques to bones, teeth and hair in particular; for example, one of HM Coroners wanted confirmation that a body, the remains of which had been found hanging in a wood, was who it was suspected to be.
Similar techniques can be used to resolve medical disputes; for instance, in a case where it was suspected that a mastectomy had been performed on the wrong woman. This was confirmed by comparing the DNA profile of a sample of the biopsy tissue taken from the woman in question with the profile of the excised breast tissue. Unfortunately, there had been a critical mix-up of samples in the hospital concerned.
Claims for compensation
We are often asked to help with claims for compensation made against manufacturers or retailers that relate to ‘contaminated’ products, especially foodstuffs. Very often the contamination involves some sort of body fluid such as blood on the wrappings or on the product itself. We regularly find ourselves examining yogurt pots, sandwiches or even, as on a recent occasion, an ice cream cone with the ice cream still inside it. In many cases, the contamination turns out to have come from the consumer – but not always. In these circumstances we may be asked to advise as to where in the supply chain the contamination could have occurred.
Contamination or sabotage of manufacturing production lines
Internally generated incidents, where disaffected or disillusioned employees relieve themselves on or into products, spit chewing gum onto them or even include sanitary tampons or used condoms in them, affect manufacturers in a very immediate way.
In one case we distinguished ourselves by obtaining a full DNA profile from urine that had been used in this way. This was a remarkable feat since urine does not normally yield much in the way of DNA-containing cellular material.
Very often the knowledge that the manufacturer has the means to identify which staff member is responsible is sufficient to persuade the guilty party to put their hands up and/or to leave, without the rest of the workforce having to have their DNA taken to be checked against the offending sample.
Hate mail and general harassment
Because it is usually difficult to handle something without leaving some trace on it, we have achieved considerable success with analysing so-called hate mail and other items sent through the post as part of campaigns of general harassment. With mail, we tend to look first for saliva on the backs of postage stamps and envelope flaps, but the process does not end there. Each case is different and may present opportunities for evidence other than DNA to be found, which enables items to be ‘clustered’ as having come from a common source. In these cases, DNA profiling tends to form just one prong of the overall investigative strategy, albeit a very important one.
Other types of case
There are many types of circumstance where DNA profiling has been instrumental in resolving suspicions or matters in dispute. One of the more unusual ones concerned a medical doctor whose excessive prescription of certain drugs had been noticed. As part of the investigation, his consulting room was searched and injection needles were recovered from several safety disposal bins. Twenty needles were submitted for examination and blood in 10 of them was sent for DNA analysis. All 10 needles generated DNA profiles that matched the doctor’s own DNA profile, and it became clear that he had been taking the drugs himself.
The essential message is that DNA profiling has a much wider application range than might be appreciated at first sight, and it is always worth asking if it might be capable of providing answers, however old or unusual the circumstances surrounding the question.